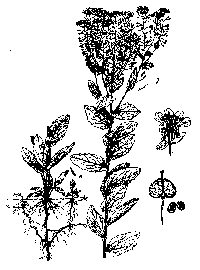|
Cardaria draba
|
|
|
|
Scientific name
|
Cardaria draba
|
|
Additional name information:
|
(L). Desv.
|
|
Common name
|
white top, perennial pepper-grass, heart-podded hoary cress, pepperwort, white-top, pepperweed whitetop, hoary cress, whitetop, white weed
|
|
Synonymous scientific names
|
Cochlearia draba, Lepidium draba, Nasturtium draba
|
|
Closely related California natives
|
0
|
|
Closely related California non-natives:
|
Cardaria pubescens
|
|
Listed
|
CalEPPC List A-2,CDFA B
|
|
By:
|
Carla Bossard,David Chipping
|
|
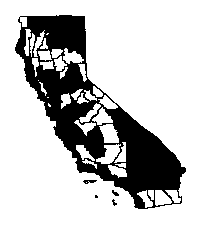
Distribution
|
|
|
HOW DO I RECOGNIZE IT?
Distinctive features:
|
Three species of hoary cress are found in
California: heart-podded (Cardaria draba), lens-podded
(C. chalepen-sis), and globe-podded (C. pubescens).
Globe-podded hoary cress is not listed as noxious in
California. These Cardaria species are single-stemmed,
upright perennial herbs, less than knee high, with upper lobed leaves clasping
the stem. The inflorescence is typically flat-topped and generally dense with
white flowers. Lower leaves form a basal rosette and are somewhat hairy and
lance-shaped; higher on the plant the leaves clasp the stem with two ear-like
lobes and have fewer hairs. Flowers have four sepals with white margins and four
petals narrowing to a claw at the base. Plants tend to form dense monospecific
mats with shoots connected by white underground rhizomes (Hick-man 1993, Robbins
et al. 1974).
åÊ
These three Cardaria species are
differentiated mainly by the shape of their fruit pods. The pods of heart-podded
hoary cress are heart-shaped at the base. The pods of the lens-podded hoary
cress are circular. The pods of both heart-podded and lens-podded are flattened
in cross-section and have no hairs. The pods of globe-podded hoary cress are
globular or spherical and are covered with fine hairs (Hickman 1993, Robbins
et al. 1974).
|
|
Description:
|
Brassicaceae. Perennial herb typically 6-20 in (15-50 cm) high. Stem: generally solitary and erect, often procumbent in older plants, hairy at the base, sparsely hairy above. Leaves: 1.5-3 in (4-8 cm) long, broadly ovate to lens-shaped, with edges entire and sometimes toothed. Basal leaves more slender but larger than stem leaves, narrow into short petiole, and form basal rosette. Stem leaves sessile, clasping stem with ear-like lobes. Leaves and stem blue-green to gray-green. Inflorescence: composed of numerous, showy, small white flowers with white 1/8 in (3 mm) petals. Flowers: in a flat top inflorescence on one to several flowering branches. There are 4 obtuse, glabrous sepals with white margins, and 4 white petals, broad at the apex and narrowing to a claw at the base. Fruit: somewhat heart-shaped, consisting of two valves and a small persistent style at the apex; becomes inflated and veined at maturity. Seeds: 0.08 in (2 mm) long, 0.06 in (1.5 mm) wide, dark reddish brown with 1-2 seeds in each half, which are finely pitted, slightly flattened, rounded at one end and narrowed to a blunt point at the other. The whitish rhizome-like rootstocks can penetrate downward several feet and laterally as far as 6 ft (1.8 m) (Hickman 1993, Cook 1987, Mulligan and Frankton 1962). |
|
|
|
WHERE WOULD I FIND IT?
|
Cardaria draba and C. chalepensis grow in many habitats and
areas of the state, except in the Mojave and Colorado deserts (Barbe 1990). C. draba occurs in wet
and dry grasslands, scrubs, and arid areas with alkali soils. It is most often
found in open, unshaded areas on disturbed, generally saline soils, but can grow
on almost any soil. It generally is found at elevations of less than 4,000 feet,
but it is known from elevations of over 8,000 feet in
Utah and over 6,000 feet in
Montana. It survives heavy frosts and snowfall, but may favor
wetter sites in harsher climates (Cook 1987, Robbins et al.
1974).
Cardaria chalepensis concentrations exist in grainfields and hayfields and
along roadsides in Siskiyou County, the Sacramento-San Joaquin Delta, and in Kings and
Tulare counties (Barbe 1990). It is particularly common in
the north on red-brown soils and in disturbed, generally saline soils and fields
(Mulligan and Findley 1974).
|
|
WHERE DID IT COME FROM AND HOW IS IT SPREAD?
|
Hoary cress is native to central
Europe and western Asia, probably centering on
Turkey, Georgia, Syria, Iraq, Iran, and Armenia. It has now spread to all continents and is
particularly common in many parts of North America.
åÊ
Cardaria species occur as crop weeds throughout the
Middle
East, Europe, Australia, and New
Zealand.
Cook (1987) reports that C. draba has been considered
EnglandÛªs most serious weed pest since 1949, and the most
serious weed pest for the Wimera region of
Australia.
åÊ
Hoary cress appeared in
New
York in 1898,
apparently introduced from ship ballast. In 1910 it was introduced into alfalfa
fields in the southwestern United
States
from imported seed from Turkestan (South Australia Dept. of Agriculture 1973; Chipping
1992). It was first reported in California near Yreka (Siskiyou County) in 1876 (Robbins 1940).
åÊ
Seed is commonly spread in hay and forage
such as cut alfalfa, in soil attached to livestock and farm equipment, and by
flowing water. Seed may be spread by wind along highways (South Australia Dept.
of Agriculture 1973; Chipping 1992). Despite prolific seed production, spread by
seed is likely not the most important means of spread. Many infestations remain
virtually the same size year after year in spite of annual seed production
(Parsons 1992).
åÊ
Plants also spread by means of extremely
persistent root systems, which consist of extensive rhizomes from which shoots
emerge (Mulligan and Findley 1974). Another method of dispersal is through
movement of root fragments in mud carried by livestock and vehicles, spread by
highway maintenance, carried in streams, and spread by tillage (Cook 1987,
Robbins et al. 1974). Even very small pieces of root are capable of
growth. Infestations in areas with frequent disturbance, such as cultivation,
regularly increase in size and density (Parsons
1992).
|
|
WHAT PROBLEMS DOES IT CAUSE?
|
Cardaria draba establishes monospecific mats that exclude most or
all other herbaceous vegetation. C. chalepensis forms dense infestations
that crowd out forage plants in meadows and fields. By displacing native
vegetation used by wildlife, both species negatively affect native fauna as
well. These Cardaria species are strong competitors for nutrients and
moisture. In AustraliaC. chalepensis slowed water drainage, increasing flooding. Gophers
increased and, as a result, the amount of soil disturbance also increased,
increasing colonization rates of Cardaria species. Australians consider
C. chalepensis toxic to grazing stock (Chipping 1972, South Australia
Dept. of Agriculture 1973).
|
|
HOW DOES IT GROW AND REPRODUCE?
|
Growth and reproduction are better understood for Cardaria draba
than for C. chalepensis, although the two may be quite similar. C. draba
reproduces by seed and expands by creeping roots. Flowering is generally from
March to June, but may occur as early as December in mild coastal climates. In
large stands that are close to the water table a few flowering plants may be
encountered year round. In early spring, infestations may resemble carpets of
snow (Cook 1987, Robbins et al. 1974, Mulligan and Findlay 1974). Under
stressful conditions flowers may develop on stems just four to six inches (10-15
cm) high with just one branch, but in well watered conditions flowering may
start when the plant is as small, but continue until many flowering branches
have developed and plant height approaches twenty inches (50 cm). The plant is
self-incompatible, is pollinated by insects, and can produce 1,000 to 5,000
seeds per stem, with seed viability of about 80 percent. Seeds are small, with
550,000 seeds per kilogram. Plants appear to produce few seeds in dry years and
are prolific seeders in wet years. Seedbanks are generally depleted in three
years under both irrigated and non-irrigated conditions. Seed survives in
uncomposted cattle dung.
|
Germination in these Cardaria species typically occurs in autumn, with
the plant overwintering as a rosette and flowering the following spring. Plants
can also germinate in spring or early summer and over-summer as a rosette, but
flowering still is delayed until the following spring. Seeds can germinate
thirty-five to forty-two days after they are released from the fruit, and in
California generally do so after the first rains. C. draba sometimes appears
after a grass fire; the heat presumably breaks the dormancy of seeds lying in or
on the soil (Parsons 1992).
|
(click on photos to view larger image)
|
|
Basal rosettes of Cardaria draba are formed three to four weeks after
sprouting. The rosettes may be formed from seed or, more commonly within
infested areas, from sprouts that arise from rootstocks. Plants usually do not
flower the first year. After twenty-five days a plant may be rooted to a depth
of 10 inches (25 cm) and may have up to six lateral roots with buds. Shallowly
buried buds may form rosettes, while deeper buds form new rhizomes.
Hoary cress has a deep, penetrating root system, numerous underground
buds, and large food reserves. The extensive root system spreads horizontally
and vertically with frequent shoots arising from the rootstock. Within three
months roots can extend a foot from the stem, with nearly fifty new shoots and
over eighty buds. In the absence of competition, one plant can produce 455
shoots the first year (Cook 1987). Energy is stored in the rootstock during the
growing season, and new plants are produced from joints in the roots. The roots
may survive complete removal of shoots for a period of one season without
noticeable loss in vigor; plants suffer visibly the second consecutive year.
Plants must be cut off at depths greater than 20 inches (50 cm) below the soil
surface to prevent regeneration from underground parts. Even root fragments will
readily regenerate, allowing C. draba to be spread by any vector that can carry
a root fragment.
|
|
|
HOW CAN I GET RID OF IT?
|
|
|
Physical control:
|
Manual/mechanical methods: Tillage may
control infestations if started at flower bud time and continued every ten days
throughout the growing season. Slightly longer intervals may be possible at
different times of year, but it is essential that no green leaves be allowed to
form. This deprives rootstock fragments of energy, but the process may have to
be continued for at least three growing seasons to deplete the seedbank. Care
should be taken not to spread fragments of the plant out of the infested area on
tillage equipment.
Prescribed burning: Cardaria
species apparently are favored by fire through removal of competition.
Flooding: For control of Cardaria
draba, flooding to a depth of six to ten inches (15-25 cm) for about three
months can produce 90 percent control of the plant (Cook 1987, Fryor and
Makepeace 1978, Pryor 1959, Robbins et al. 1974). However, short-term
submergence lasting a week has no effect on the plant (Chipping, pers.
observation).
|
|
Biological control:
|
Insects and fungi: No USDA recommended
biological control agents exist, and potential introductions from the native
range are complicated by the large numbers of cruciferous crops. Although the
mite Acerea draba is effective in sterilizing plants of Cardaria
draba, it is also found on commercial crops, as is the aphid, Aphis
armoracea (Cook 1987).
åÊ
Grazing: Grazing is not effective on
Cardaria draba, as it survives and resprouts using energy stored in its
rhizome-like rootstock. In C. chalepensis young plants may be grazed to
the ground by cattle and sheep, which also ingest seed heads.
Although C. chalepensis contains
glucosinolates and can be mildly toxic, nutritional levels are adequate to meet
the requirements of most livestock, especially in early growth stages. Problems
arise as the foliage becomes coarse and bitter as it matures, when plants have
low nutritive value compared to other forages (Cook 1987, Robbins et al.
1974).
åÊ
These plants actually are spread by
grazing, as cattle ingest seed heads and may become vectors for plant
fragments.
|
|
Chemical control:
|
Most research on chemical control of Cardaria
species has focused on cropland, usually alfalfa, clover, or wheat fields.
Experiments commonly include combinations of herbicides and other non-chemical
methods in association with the herbicide treatment. Check with your county
agricultural agent to determine which of the possible chemical means of control
are currently registered for use in wildlands in California.
Different forms of 2,4-D have been tried with limited
success in northern California, although Canadian trials have had success with
applications at 1 to 2 lbs/acre, repeated for three years to remove the
seedbank.
Mixes of 2,4-D ester and dicamba have
been applied by aircraft, and mixes of 0.50 2,4-D and 0.25 each dicamba and R-11
surfactant have worked in roadside applications of one gallon of the mix in 100
gallons of water. Airplane application inevitably affects non-target plants and
carries with it the danger of drift of the herbicide to non-target areas and
surface water.
Chlorsulfuron,
which is selective for broadleaf plants, has been used on
California
rangeland at 0.33-1 oz/acre with limited success, but has a half-life of four to
six weeks and affects non-target species. Chlorsulfuron at 0.50-2 oz/acre has
been successful in roadside applications in central coastal
California.
Glyphosate at 1 pt/acre produces 80 percent control at the budding or flowering
stage, but is also non-selective (Cook 1987, Fryor and Makepeace 1978, Pryor
1959, Robbins et al. 1974).
|