|
Ficus carica
|
|
|
|
Scientific name
|
Ficus carica
|
|
Common name
|
edible fig, common fig
|
|
Synonymous scientific names
|
none known
|
|
Closely related California natives
|
0
|
|
Closely related California non-natives:
|
0
|
|
Listed
|
CalEPPC List A-2,CDFA nl
|
|
By:
|
John M. Randall
|
|
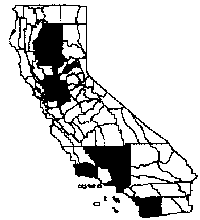
Distribution
|
|
|
HOW DO I RECOGNIZE IT?
Distinctive features:
|
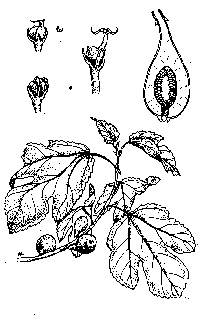
Edible fig (Ficus carica) is the familiar
fig tree that produces edible fruits sold fresh, dried, and as paste used as
filling for cookies (fig newtons) and other sweets. Mature trees often have
multiple trunks and may grow to thirty feet tall. The heavy trunk and branches
are covered with a smooth, light gray, flaky bark. The sap is thick, sticky, and
slightly milky. The leaves are rough to the touch, bright green, with three to
five lobes, the classic fig-leaf shape. They are two to eight inches wide and
two to ten inches long with a two- to four-inch-long petiole (stem). Edible figs
are deciduous, dropping their leaves in mid- to late autumn and leafing out
again in early spring (March-April). The fruits are shaped like small sacks full
of sand, two to four inches long and nearly as wide. Edible fig has been widely
cultivated, and the color of ripe fruits of different cultivars ranges from dark
purple-black in the Û÷MissionÛª fig to pale greenish yellow in the Û÷KadotaÛª and
Û÷GenoaÛª figs to white with a purple blush in Û÷ConadriaÛª figs. Cultivated trees
produce large numbers of fruits, which can become a nuisance when they ripen and
drop, but those growing wild in canyons and riparian (riverside) forests around
CaliforniaÛªs Central Valley usually produce only a few greenish yellow fruits
that ripen in late summer or fall.
åÊ
|
|
Description:
|
Moraceae. Tree up to 30 ft (10
m) tall, winter-deciduous, leaves dropping in mid- to late autumn and emerging
again in early spring (March-April) in California. Leaves with petioles
| The multiple fruits of caprifigs are dry and chaffy and regarded as unpalatable, while those of figs are fleshy and savored by many birds and mammals, including humans. Botanically speaking, the fleshy outer tissue of the fig is actually the receptacle, while the true fruits, enclosed within the receptacle, are single-seeded drupelets with hard, thin outer walls that give figs their characteristic crunchy texture (description from Hickman 1993, Lisci and Pacini 1994). |
|
|
|
WHERE WOULD I FIND IT?
|
Edible figs invade and dominate riparian forests, streamside
habitats, levees, and canal banks in and around CaliforniaÛªs Central Valley,
surrounding foothills, the south coast, and the Channel Islands (Hickman 1993).
They are also widely cultivated for fruit and ornament in areas below 2,500 feet
(800 m) elevation. California is one of the worldÛªs largest producers of fig
fruits (Ferguson et al. 1990).
Edible fig is most likely to escape where soils stay moist
throughout the summer. It has invaded many nature preserves and parks in
California. Plants form dense thickets covering roughly twenty-five acres along
a seven-mile-long section of Dye Creek at the Dye Creek Preserve northeast of
Chico and have begun to invade the riparian forest at Woodson Bridge State Park
along the Sacramento River to the west. Several rapidly expanding fig thickets
were found in the most pristine valley oak riparian forest on the Cosumnes River
Preserve south of Sacramento. These thickets were repeatedly cut and the stumps
treated with herbicide, but they were difficult to eliminate. Edible figs have
also invaded parts of the riparian forest in Caswell State Park near Stockton.
They are found on the Santa Cruz Island Preserve in disturbed sites, and
scattered along coastal flats and in coastal scrub (Junak et al. 1995).
åÊ
|
|
WHERE DID IT COME FROM AND HOW IS IT SPREAD?
|
Edible fig is probably
native to the fertile region of southern Arabia (Ferguson et al. 1990). It was
probably first domesticated outside its native region, in Mesopotamia, in the
valley of the Tigris and Euphrates rivers, in what is today Iraq. It was among
the earliest known fruits to be cultivated, with records dating back to the
Sumerian era, roughly 4,900 years ago (Ferguson et al. 1990). The area where
figs were cultivated gradually grew to encompass all nations of southwestern
Asia and the Mediterranean Basin. Edible fig was first introduced to the New
World in the West Indies by Spanish and Portuguese missionaries in the 1520s and
then to what is now the East Coast of the United States in 1575 (Ferguson et al.
1990). Edible fig trees were introduced to California by Spanish missionaries
beginning in 1769; hence this variety is known as Û÷MissionÛª or Û÷FranciscanÛª fig.
Many other varieties were introduced to California for food and ornamental
plantings after 1850. Commercial production of figs was hampered until the fig
wasp, Blastophaga psenes
, was successfully introduced to the state in 1899 (Ferguson
et al. 1990). Currently, California ranks third in the world in fig production,
behind Turkey and Greece, although figs rank only twenty-second in value and
eighteenth in acreage among crops produced in California (Michailides et al.
1996).
It is not clear how edible fig spreads into preserves and other
wild areas. It grows quickly and can spread vegetatively by root sprouts, soon
forming dense thickets that exclude most other plants. Limbs that have been cut
or broken and fallen to the ground can take root, and it is thought that
branches broken off during storms or floods may wash up and root at downstream
sites. Many birds eat the fruits and may spread the seeds. Hujik (pers. comm.)
reports that deer also feed on the fruits. Seeds germinate only if they are
removed from the fleshy synconium during passage through an animalÛªs gut or by
mechanical means such as heavy rainfall (Lisci and Pacini 1994).
åÊ
|
|
WHAT PROBLEMS DOES IT CAUSE?
|
If not controlled, edible fig trees could
crowd out native trees and understory shrubs characteristic of CaliforniaÛªs
riparian forests. Riparian forests are already rare in California, especially in
the Central Valley, where over 95 percent have been converted to cropland,
pasture, or developed areas in the past 150 years. No published or unpublished
reports are available with quantitative information on the impacts of edible
figs invading natural vegetation in California or elsewhere.
The leaves of edible fig contain at least two furocoumarin
compounds that are activated on exposure to a certain waveband of light and can
then cause a skin rash in humans (Damjanic and Akacic 1974, Evans and Schmidt
1980). The activated furocoumarins are primary irritants, meaning they
chemically or mechanically irritate the skin rather than causing an allergic
response. The mode of action of these compounds is not known, but they may
photobind to DNA and/or ribosomal RNA in epidermal cells following exposure to
ultraviolet light in the 320-370 nm waveband (Evans and Schmidt
1980).
åÊ
|
|
HOW DOES IT GROW AND REPRODUCE?
|
Most edible fig fruits with viable seeds are produced in late summer and in
autumn. Studies in Europe indicated that, once freed from the fleshy synconium, edible fig seeds may germinate in autumn or in spring, depending on climatic conditions (Lisci and Pacinia 1994), and the same presumably is true in California. Seeds germinate at temperatures between 50 and 85 degrees F (10 – 30 degrees C), but only if humidity remains high or if they are in contact with soil that is continuously wet (Lisci and Pacini 1994).
Edible fig grows quickly in soils with enough moisture and with
exposure to high light levels. It is winter-deciduous, and the timing of
leaf-out and leaf drop varies with the cultivar and with climatic conditions.
Edible fig reproduces by seed and by vegetative growth. Most of
the worldÛªs Ficus species depend on a species-specific agaonid wasp
(family Agaonidae, Hymenoptera) for pollination. Ficus carica depends
on the wasp Blastophaga psenes. The wasps are in turn dependent on
F. carica because they breed only inside its fruits (Kjellberg et al.
1987). Fertilized female B. psenes wasps squeeze through the
scale-covered ostiole in the end of the synconia of caprifigs and lay one egg in
each of several of the short-styled female flowers. Each wasp larvae destroys a
female flower as it feeds and grows. Female flowers that escape the egg-laying
may each produce a single viable seed if pollinated. Adult male wasps emerge
first and quickly cut into flowers containing female wasp larvae and mate with
them. These female wasps emerge two to three weeks later and, as they make their
way out of the synconium, they pick up pollen from the male flowers clustered
near the opening. They then search for another, younger synconium and squeeze
through the narrow opening to reach the flowers inside. Galil and Newman (1977)
found that the opening is so small that it is likely all pollen on the body
surface of the wasp is scraped off as it squeezes through. They speculate that
the wasps carry pollen in cavities on their bodies (e.g., between abdominal
segments) and that these pollen grains fall out and onto fig flowers when the
waspÛªs body swells and twists as it attempts to deposit eggs. The wasps insert
their ovipositors down the style tube to deposit their eggs, but the styles of
the female flowers in fig synconia are so long that they prevent the wasps from
successfully depositing eggs. During their exertions, however, the wasps deposit
pollen and fertilize these flowers. The undamaged, pollinated long-styled female
flowers develop into tiny seeds within the synconia. Seeds from these flowers
can produce both types of tree, caprifigs and figs (Beck and Lord 1988). Once a
female B. psenes wasp has entered a synconium to lay eggs, it cannot leave, so
these individuals are destined to die without producing offspring (Kjellberg et
al. 1987).
In California caprifigs produce three crops of fruit, one each
in winter, spring, and summer. If climate allows, figs may produce two crops, a
small one initiated in spring and maturing in June or July and a main crop
initiated between May and July and ripening between August and December
(Ferguson et al. 1990). Staggering of fruit crops enables the fig wasp to
survive throughout the year and ensures pollination of flowers in fig synconia.
The synconia of the main crop of Smyrna varieties of edible fig, including the
Û÷CalimyrnaÛª cultivar, will abort and fail to develop if flowers are not
fertilized. Synconia of common figs, including the Û÷Mission,Ûª Û÷Adriatic,Ûª and
Û÷KadotaÛª cultivars, can develop fully even if the flowers are not pollinated and
no viable seeds are produced (Ferguson et al. 1990).
Because the wasps can carry fungal disease spores, California
fig growers often grow caprifigs apart from fig trees and distribute ripe
caprifig synconia with emerging wasps around their orchards only after checking
them for disease (Michailides et al. 1996). It is not known whether the edible
fig trees that have invaded natural areas in California are all of one variety,
nor is it known whether the individuals are caprifigs (with male and
short-styled female flowers) or figs (with only long-styled female flowers).
The fleshy tissue of the synconium apparently contains
inhibitors and/or creates a microenvironment with high osmotic pressure that
prevent seeds from germinating (Lisci and Pacini 1994). Birds and mammals feed
on and pick apart the fig fruit and then excrete or drop seeds, releasing them
from this inhibition and usually distributing them as well. Seeds may also be
washed free of the synconium by hard rains after they fall to the ground and
split open.
|
Edible fig may begin to produce fruit (synconia) within one year if propagated by cuttings or within two to three years if propagated by seed under favorable conditions in orchards. Numbers of fruit are small the first few years, but orchard plantings usually bear harvestable crops by their fifth year.
|
(click on photos to view larger image)
|
|
|
HOW CAN I GET RID OF IT?
|
An efficient control
method for edible fig has not yet been developed. The trees resprout vigorously
after cutting and are difficult to control without herbicides.
|
|
Physical control:
|
Manual/mechanical: Edible figs are
shallow-rooted in heavy, wet soils typical of riparian forests and can be pulled
up fairly easily when young. They often root-sprout, however, so that what looks
like one small sapling may be one of many sprouts from a large network of roots.
A small or medium-sized weed wrench may help remove some of the mid-sized
specimens. Repeated cutting of resprouts may eventually exhaust the root
reserves of a tree or small thicket if the interval between cuttings is short
enough, but this has not yet been demonstrated.
åÊ
|
|
Biological control:
|
Insects and fungi: No biological control species are approved by the USDA for this species. However, figs are subject to
damage from nematodes, tree borers, and rust.
|
|
Chemical control:
|
At the Cosumnes River Preserve all trunks
and sucker shoots in a thicket were cut six to eighteen inches above the ground
and the cut stumps treated with a 100 percent solution of an amine formulation
of triclopyr (sold under the names Garlon3Aå¨ and Brush-B-Goneå¨). This was
successful, although some thickets had to be retreated at least once because
there was some resprouting. The retreatments were carried out at yearly
intervals, but shorter intervals (two to six months) might have improved their
impact by giving the plants less time to replenish root reserves. Managers at
the Cosumnes River Preserve recently have been using a hack-and-squirt method,
applying 100 percent triclopyr amine formulation to the wounds, but it is too
early to tell if this will be as effective as the cut-stump treatments. This
method was also tried at the Dye Creek Preserve, but was not effective there.
Herbicide may be applied in an eight- to twelve-inch-wide band
around the uncut trunks of trees with trunk diameters up to two or three inches
and perhaps greater. This is known as basal bark application, and it has been
shown to be highly effective for a variety of trees and shrubs. Other
herbicides, including glyphosate (marketed under a variety of names, including
Rodeoå¨ and Roundupå¨) and imazapyr (as Chopperå¨ and Arsenalå¨) may be at least as
effective as triclopyr against edible fig, but studies of this have yet to be
conducted.
åÊ
|