|
Schinus terebinthifolius
|
|
|
|
Scientific name
|
Schinus terebinthifolius
|
|
Additional name information:
|
Raddi
|
|
Common name
|
Brazilian pepper tree, Christmas-berry tree, Christmasberry, Florida holly
|
|
Synonymous scientific names
|
Schinus mucronulata, S. antiarthriticus
|
|
Closely related California natives
|
0
|
|
Closely related California non-natives:
|
Schinus molle
|
|
Listed
|
CalEPPC List B,CDFA nl
|
|
By:
|
Johathan Randall
|
|
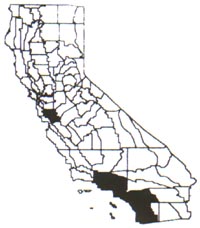
Distribution
|
|
HOW DO I RECOGNIZE IT?
Distinctive features:
|
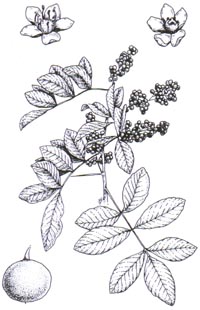
Brazilian pepper tree (Schinus
terebinthifolius) is a many-stemmed shrub or small tree. Its branches form
a nearly impenetrable tangle down to ground level. The flowers are small, less
than one-tenth of an inch in diameter, and have five green-tinged, white petals.
The tree is covered with round, bright red fruit from December to February. The
odd-pinnate compound leaves have a narrowly winged rachis and rounded, often
toothed leaflets that give off a strong smell of turpentine when crushed
(Tomlinson 1980).
åÊ
|
|
Description:
|
| Anacardiaceae: Shrub or small tree to 40 ft (13 meters) tall with a multi-stemmed short trunk that is curved with grayish and often scaly bark. Leaves: 0.32-0.6 in (8-15 cm), generally opposite, with a somewhat resinous coating and emitting a distinctive odor. Usually 7-9 leaflets, odd-pinnate, sessile or short-stalked, 1-2.8 in (2.5-7 cm) long, 0.4-1 in (10-25 mm) wide, elliptic to oblong, with edges entire to toothed. Inflorescence: panicle, either axillary or terminal. Flowers: 0.04-0.09 in (1.2-2.5 mm) long, with greenish white, oblong to egg-shaped petals and pedicels 0.08-0.16 in (2-4 mm). Fruit: drupes, 0.16-0.28 in (4-7 mm) in diameter, generally pink to red, arranged in dense bunches (Ewel et al. 1982). Each drupe contains a single seed (Ewel et al. 1982). |
|
|
|
WHERE WOULD I FIND IT?
|
While it can be found in Santa Clara County
in northern California, Brazilian pepper tree is far more common and problematic
in southern California from Riverside to the coast, including Ventura and San
Diego counties. Usually it is found below 200 meters elevation, especially in
canyons and washes (Hickman 1993). Brazilian pepper is capable of surviving a
broad range of hydrologic conditions, but does best in well drained sites (Ewel
1979). This species is a pioneer of disturbed sites, such as highway
rights-of-way, fallow fields, and drained bald cypress stands. It can also
establish in undisturbed wildlands (Woodall 1982). It causes severe problems in
southern Florida, where it invades pinelands, mangrove forests, and marshes, and
it is a major invader in croplands under restoration in Everglades National
Park.
åÊ
|
|
WHERE DID IT COME FROM AND HOW IS IT SPREAD?
|
Brazilian pepper tree is native to
Argentina, Paraguay, and Brazil, where it occupies dry savannah (Nilsen et al.
1980). It is a sparsely distributed species in its native range, where it rarely
acquires the dominance that it achieves in California and other areas in which
it has become naturalized. It was introduced into Florida in 1891 as an
ornamental, although there is evidence that it arrived in the United States
fifty years earlier (Gogue et al. 1974). It is still used as an ornamental in
California, Texas, and Louisiana. Other than escaping from human cultivation in
gardens, Brazilian pepper tree spreads via small mammals and birds, especially
robins, which eat the drupes and then distribute the seeds during their travels.
Mammals such as raccoons and possums consume the fruits and deposit the seeds
with fecal materials, giving the seeds a nutrient-rich microsite in which to
establish (Ewal et al. 1982). This species has infiltrated the islands of
Hawaii, Bermuda, and the Bahamas, as well as Florida and, more recently,
California. In addition to spreading by seed dispersal, it can resprout,
especially after fire (Elfers 1995).
åÊ
|
|
WHAT PROBLEMS DOES IT CAUSE?
|
Outside its native home, Brazilian pepper
tree spreads aggressively. Typically, dense monospecific stands form within a
few years after trees invade an area. This often creates a dense canopy and can
shade out most competing vegetation, posing a serious threat to natural
vegetation and organisms that depend on them (Bennett et al. 1988, Doren and
Whiteaker 1990, Ewel 1979). The tenacity of Brazilian pepper seedlings impairs
competition by native vegetation. Brazilian pepper also seems to produce
allelopathic chemicals in the soil that inhibit growth of other plants (Bennett
et al. 1988). The plant is moderately salt tolerant, withstands flooding, fire,
and drought, and resprouts quickly after being cut. Its ability to resprout and
grow rapidly allows it to quickly dominate burned areas.
åÊ
|
|
HOW DOES IT GROW AND REPRODUCE?
|
Brazilian pepper tree reproduces by seed. Flowering begins in September, and by mid-October almost every tree is in flower. Most flowering ceases in early November. A small fraction of the population flowers in March-May. Fruit ripening follows close behind flowering, with most occurring between December and February (Ewel et al. 1982). Mature female trees are prodigious seed producers, which, combined with a viability rate of 30 to 60 percent, results in a vast number of seedlings (Elfers 1995). It has been suggested that female flowers may
mimic male flowers, attracting foragers in search of pollen (Ewel et al. 1982). Fruit production occurs in winter (November to February), at which time the branches of female trees are heavily laden with red fruits.
Ripe fruits are retained on the tree for up to eight months, and
all are dispersed before the next flowering season (Ewel et al. 1982). The
attractive fruits are readily eaten and transported by birds and mammals, with
water and gravity serving as less important dispersal agents. Seed dispersal by
native and exotic birds, such as catbirds, mockingbirds, American robins, and
red-whiskered bulbuls, accounts for the presence of Brazilian pepper tree in
almost every terrestrial plant habitat in southern Florida (Austin 1978, Ewel et
al. 1982, Ewel 1986). Robins, when present, are believed to consume and
transport more Brazilian pepper tree seed than all other dispersal agents
combined. Raccoons and possibly possums are known to ingest the fruits (Ewel et
al. 1982). The fact that little else is fruiting during the winter months when
Brazilian pepper tree seeds are dispersed has been suggested as a possible
explanation for the success of this plant in southern Florida (Ewel 1986).
Seeds are generally not viable beyond five months after
dispersal. However, Ewel (1979) reported seed germination in late fall. Under
certain conditions; seeds apparently retain their viability during the wet
season and germinate when water levels drop later in the year.
Most seed germination takes place between January and February,
but the range is from November to April. Seeds germinate within about twenty
days. Seedlings have been found to grow quickly in most young successional
communities and slowly in most older communities. Well watered conditions favor
germination. Survival of Brazilian pepper tree seedlings, even of mature forest
trees, is 66 to 100 percent (Ewel 1986). Ewel et al. (1982) concluded that the
growth plasticity of Brazilian pepper tree seedlings makes this species
especially difficult to manage. Seedlings can grow slowly under the dense shade
of mature stands and then exhibit vigorous growth when the canopy is opened
after a disturbance. In exposed, open areas, such as young successional
communities, their growth rate is as high as twelve to twenty inches (30-50 cm)
per year.
Under favorable growing conditions, Brazilian pepper tree can
reproduce within three years after germination. This species can occur
individually or as extensive stands; in areas with warm, tropical conditions, it
occurs primarily in dense stands.
|
Like many hardwood species, Brazilian pepper tree can resprout from above-ground stems and root crowns following stump cutting, bark girdling, fire that girdles a stem, and herbicide application (Woodall 1979). Resprouting is often profuse, and growth rates of sprouts, which originate from dormant and adventitious buds, are high. Brazilian pepper treeÛªs generally shallow root system also favors the production of underground root suckers. Root suckers form without evidence of damage to a tree or its root system and can develop into another plant. The clumping of trees often seen during the early stages of invasion can be explained by this suckering mechanism (Woodall, 1979).
|
(click on photos to view larger image)
|
|
|
HOW CAN I GET RID OF IT?
|
The severity of the problem is an important
consideration when designing a control strategy for Brazilian pepper tree. In
many cases, it is unrealistic to attempt eradication where this species is
widespread; instead the goal should be to prevent colonies from establishing new
infestations. Because this species is an invasive, habitat-altering weed in
other states, especially Florida, research on control techniques has been
underway for some time (Elfers 1995). With all control and removal techniques, a
lack of sprouting for one or even two years may not guarantee that the sprouting
potential of the roots is exhausted. Yearly monitoring for at least three years
following control efforts is recommended. Monitoring should occur in late spring
to determine if any shoots have survived or any resprouting has occurred in
treated areas.
åÊ
|
|
Physical control:
|
Manual/mechanical methods: Entire saplings, including root
systems, can be pulled up by hand, but by the time the plant is several feet
tall, hand pulling may be impossible. If as much as one-quarter of the root
system is left in the ground, the plant may resprout. Using heavy equipment such
as bulldozers, the entire plant, including the root system, can be removed.
Soil removal: A study in south Florida found that removing soil
with a bulldozer down to the limestone bedrock effectively prevented the return
of Brazilian pepper tree. It also stimulated the growth of a diversity of plant
species. However, this site had little or no soil before it was rock-plowed and
farmed, so removing the soil was part of a logical restoration program. Removing
soil can also stimulate the growth of non-natives that thrive in disturbed
areas.
Prescribed burning: Results of burning were mixed. Because they
cannot tolerate heat, Brazilian pepper tree seeds will not germinate following
fire, although basal trunk and root sprouting is aggressive. Once saplings
attain a height of one meter, most are able to survive fire by regrowth from the
roots. Since it can grow more rapidly than competing native hardwoods, Brazilian
pepper tree can establish or reestablish dominance (Loope and Dunevitz 1981).
Repeated burning does not prevent this plant from reinvading an area in which it
was previously dominant (Doren and Whiteaker 1990).
Flooding: Prolonged submergence may result in increased
seedling mortality (Ewel et al. 1982).
åÊ
|
|
Biological control:
|
Insects and fungi: No USDA biological
control agents have yet been approved by the USDA, but research is underway,
especially in the heavily infested states of Florida and Hawaii. In 1988 a
seed-eating wasp, Megastigmus transvaalensis, was reared on Brazilian pepper
tree fruit collected from Palm Beach, Florida. Part of the controversy
surrounding the development of plant-eating fauna for this species is the
concern that insects that attack Schinus terebinthifolius will also attack the
closely related S. molle, a common ornamental. A study in Hawaii reported that a
caterpillar, Epsismus utilis and a beetle, Bruchus atronotatus, had the
potential to limit seed production, but field tests found that these insects did
not significantly control the plant (Clausen 1978).
åÊ
|
|
Chemical control:
|
Triclopyr (as Garlon 3-Aå¨), applied at 100
percent and using the frill-cut method, has been shown to kill mature trees and
prevent regrowth. Depending on the setting, it may be advantageous to cut down
the trees down and then apply herbicide to the stumps to prevent regrowth (Bily,
pers. comm.).
Successful treatments for full-sized plants also include basal
spot applications of bromacil and hexazinone, which kill by blocking
photosynthesis. For widely scattered plants, where access to the main stem is
difficult, basal spot treatments are easily applied and effective. Bromacil and
hexazinone are selective, so nearby vegetation is not harmed. Since Brazilian
pepper treeÛªs transpiration rate per unit leaf area is unusually high, and since
it generally occupies an emergent canopy position, it acts as a strong sink for
soil-applied herbicides, thus minimizing leaching losses and off-target damage.
When it is growing in the shade of other plants, this generalization does not
hold (Woodall, 1982).
Foliar herbicides are the fastest acting
with the least residual activity, although their probability of success is
relatively low. Foliar herbicides that have been used with some success include
15 percent triclopyr (as Garlon 3Aå¨) diluted with water or diesel, 2 percent
triclopyr (as Garlon 4) diluted with water, and 2 percent dicamba (as Banvel
720å¨) diluted with water. Ammonium-sulfate and glyphosate are also foliage
absorbed. All of these herbicides are most effective when applied to seedlings.
The only herbicide that resulted in consistently killed roots as well as shoots
was picloram (Woodall 1982), but this is not currently registered for use in
California.
åÊ
|