|
Cynara cardunculus
|
|
|
|
Scientific name
|
Cynara cardunculus
|
|
Common name
|
artichoke thistle, cardoon, desert artichoke, wild artichoke
|
|
Synonymous scientific names
|
none known
|
|
Closely related California natives
|
0
|
|
Closely related California non-natives:
|
Cynara scolymus
|
|
Listed
|
CalEPPC List A-1,CDFA B
|
|
By:
|
Michael Kelly
|
|
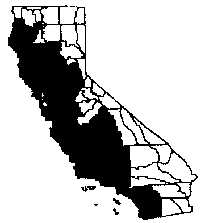
Distribution
|
|
|
HOW DO I RECOGNIZE IT?
Distinctive features:
|
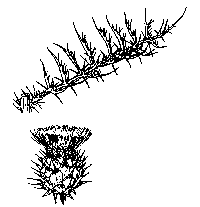
Artichoke thistle (Cynara
cardunculus) is a spiny thistle of the sunflower family, head high,
occasionally taller, crowned by a cluster of showy, bright purple thistle
flowerheads that are two to three inches in diameter from April through July.
Rarely, patches of white-flowering plants are found (Munz 1963, Kelly pers.
observation). One to several stout flower stalks rise from a bushy rosette up to
five feet in diameter. A spray of basal leaves, each deeply lobed and
gray-green, to four feet in length, arches gracefully up and out from the base.
Smaller leaves, otherwise similar in appearance, grow from the flower stalk as
it extends upwards. Stout spines on the leaves, stems, and bracts around the
flowerheads make it easy to recognize.
åÊ
|
|
Description:
|
|
Compositae. Perennial herb. Stems: leafy, branched, stout, and generally erect unless heavy flowerheads cause bending, to 5 ft (1.5 m) in height. Leaves: basal until bolting, at which time cauline leaves appear off inflorescence stalk, alternate, 1-4 ft (3-12 dm), pinnately lobed to divided, very spiny. Gray-green upper surface loosely cobwebby, lower surface densely gray-tomentose. Rubbing leaf surface easily removes cobwebby material. |
|
Inflorescence: lower heads typical of aster
family, discoid, large, with one to several per cyme in a loose cluster at top
of stalk. Typically, the uppermost flower opens before lower or peripheral
flowers on any given axis. Involucres: ovoid or hemispheric, 1-2.25 in (3-6 cm)
in height, 1.5-2.75 in (4-7 cm) diameter, not including tips, tending to narrow
above. Phyllaries surrounding flowerhead easy to identify, with stout spines at
tips, overlapping in series, generally ovate, leathery, entire, glabrous,
receptacle flat, fleshy, bristly. (Spiny tips on phyllaries on immature
flowerhead easily differentiate this plant from the closely related Cynara
scolymus, the common agricultural globe artichoke.) Flowers: striking, with
purple, sometimes blue corollas, +/- 2 in (5 cm), tube very slender, throat
widened abruptly, lobes linear; anther bases long-sagittate, tips oblong; style
appendage long, cyclindric, minutely papillate, tip barely notched. Fruits
cylindric to obconic, =/- 4-angled or +/- compressed, glabrous, attached at
base; pappus of many stiff bristles, 1-1.5 in (2.5-4 cm), in several series,
white or brownish, plumose below, fused and falling together (Hickman 1993).
Corollas occasionally white (Munz 1963, Kelly pers. observation).
åÊ
|
|
WHERE WOULD I FIND IT?
|
Artichoke thistle is found in disturbed
places, to 1,650 feet (
Artichoke thistle has been observed colonizing riparian
woodlands and natural openings in chaparral and coastal sage scrub, growing
under willow, mulefat, and sycamore, as well as in native grasslands (Pepper and
Kelly 1994). It does well in soils with a heavy clay content (Thomsen et al.
1986), which helps explain its invasion of grassland habitat occupied by
endangered San Diego thornmint (Acanthomintha ilicifolia) (Kelly
1996).
åÊ
|
|
WHERE DID IT COME FROM AND HOW IS IT SPREAD?
|
Native to the Mediterranean (Hickman 1993), artichoke thistle
became widespread on over 150,000 acres of California rangeland and also in
Australia, New Zealand, and South America on grazing lands, especially the
Argentine pampas (Thomsen et al. 1986). It is now recognized as the wild form of
the cultivated globe artichoke, Cynara scolymus L. When grown from
divisions of the perennial crown, globe artichoke will reliably produce the
spineless, edible flowerhead and plant known to agriculture, but grown from seed
it often reverts to a wild form, producing the inch-long spines around the
flowerhead normally found on C. cardunculus (Thomsen et al. 1986).
Artichoke thistle was known as the ÛÏedible thistleÛ and
mentioned in literary references dating back to several centuries before Christ.
Cultivation of the edible wild form appears to have spread from Naples, Italy,
into the broader Mediterranean during the fifteenth century (Warren 1996).
Cultivated forms apparently were developed from artichoke thistle in monastery
gardens during the Medieval period (Thomsen et al.1986). Emigrants from the
Mediterranean region apparently carried one or more forms of the plant to other
countries. During his voyage on the Beagle, Darwin (1989) found artichoke
thistle had already reached the Argentine pampas and escaped cultivation: ÛÏVery
many, probably several hundred square miles are covered by one mass of these
prickly plants and are impenetrable by man or beast. Over the undulating plains
where these great beds occur, nothing else can now live.Û
Botanical surveys in California from 1860 to 1864 reported the
globe artichoke variety as having escaped cultivation. Artichoke thistle was
already reported as having established itself outside cultivation in a pasture
in San Diego County in 1897. Its appearance in California rangelands can be
traced to its introduction for ornamental and culinary uses (Thomsen et al.
1986).
Although cultivated vegetatively from crown division, artichoke
thistle in the wild spreads only by seed. The large seeds are dispersed by a
variety of mechanisms. Bristles from the receptacle and the pappus (thistledown)
remain attached to the seeds when they are released, aiding in dispersal. Seeds
separate easily and quickly from the thistledown and, because of their large
size and weight, they usually are not carried more than sixty-six feet (20 m)
from the parent plant. Strong winds, however, may pile up great banks of
thistledown against fence lines and road margins. However limited this dispersal
mechanism may be, over a few years wind can significantly expand the patch size
of a local infestation.
Most seeds seem to fall close to the parent plant. Birds feeding
on the seed heads probably knock some seed to the ground and occasionally move
it greater distances. Hillside patterns suggest that water and gravity carry
seeds short distances on slopes. Seeds may attach to cattle and other mammals,
and the plant is known to spread along game trails in coastal sage scrub in
southern California. Its distribution along utility roads in some areas of San
Diego County suggests that vehicle tires are transporting seeds.
åÊ
|
|
WHAT PROBLEMS DOES IT CAUSE?
|
Artichoke thistle is an important rangeland
problem because it reduces forage production and limits movement of livestock
(Thomsen et al. 1986). The stout, upright yet spreading nature of the plant, its
formidable spines, and high densities make wildlife movement through it
difficult. The arching leaves shade a considerable area. Combined with its
aggressive root system, artichoke thistle outcompetes native vegetation for
light, water, and nutrients. At high densities it becomes a monoculture that
excludes shrubs, herbaceous plants, and even annual grasses. For example, mature
broom baccharis (Baccharis sarothroides) declines in vigor when in close
proximity to artichoke thistle. The thistle also is a threat to the endangered
San Diego thornmint.
There appears to be no alteration of soil chemistry or
allelopathy, since other species, including those originally displaced, readily
recolonize the site once artichoke thistle is dead. It is unlikely that the
plant alters fire cycles, since it mainly displaces annual exotic grasses, which
are highly combustible. However, one 4,000-acre infestation in San Diego was
first colonized by this thistle after a grassland fire in the same area in the
mid-1980s (Dumka 1997).
åÊ
|
|
HOW DOES IT GROW AND REPRODUCE?
|
Artichoke thistle flowers as early as April in southern California and into July throughout its range. In a year of average rainfall in San Diego of nine inches (22.5 cm), a mature plant can produce more than a dozen flowerheads with as many as 200 seeds per head (Kelly 1996). In drier years most established plants sprout, flower, and produce seed, but these plants are usually smaller than plants growing during wetter years. Some established plants will sprout and then wither before flowering.
|
Bees, a common visitor to the flower, are probably the most important pollinator. Mature plants resprout repeatedly when cut or when herbicide is applied too early or in too small amounts (Pepper and Kelly 1994).
|
(click on photos to view larger image)
|
In southern California artichoke thistle
germinates as early as December and as late as July. Seven years of control
efforts with careful record keeping in Pe̱asquitos Canyon Preserve (San Diego)
suggests that a sizable seedbank with an average duration of five years can be
expected (Kelly 1991). In drier years seedling mortality is high. Growth is
rapid during cool and wet winter months in coastal and southern California.
Plants usually flower the first year if rainfall is equal to or greater than
average, but flowering may be delayed until the second summer in drier years.
Late-germinating plants barely reaching a foot in height are capable of
producing flowers and setting seed. Plants form multi-stalked clumps after the
first year and the clumps enlarge thereafter. A deep taproot develops during the
first year (Parsons 1992). The roots can eventually reach eight feet in depth.
Rosette leaves usually die over the summer. The plant tends to produce
single-species stands that can reach densities as high as 22,000 plants per acre
(Thomsen et al. 1986). With a large, expanding underground taproot and tuber, a
single plant quickly becomes a many-stemmed clump. Some populations become dense
enough to restrict growth, resulting in tall, spindly plants rather than the
robust, broad clumps typical of plants not under competitive
pressures.
åÊ
|
|
HOW CAN I GET RID OF IT?
|
Because of artichoke thistleÛªs ability to
resprout after chemical spraying and to build up a seedbank that lasts five
years or more, yearly monitoring and repeat eradication are necessary.
Eradication is most effective when mature plants are bolting, generally in early
to mid-April in southern California and late April or May farther north.
Chemical control efforts have proved successful on sizable populations in
several open-space parks and on military lands managed for their natural
resources in San Diego County. Seedbanks in some areas have been exhausted, and
maintenance is minimal and routine (Kelly pers. observation).
Historically, artichoke thistle was controlled by hand grubbing,
root-plowing by tractors equipped with a specially designed blade, displacement
planting, and applying the herbidide 2,4-D from airplanes and helicopters. These
methods often proved too labor-intensive or expensive to be used successfully in
larger infestations. Costs of control efforts sometimes exceeded the value of
the land (Thomsen et al.1986). Workers often wear chainsaw chaps as protection
when working to control it.
åÊ
|
|
Physical control:
|
Manual methods: Grubbing is practical when only scattered plants
are present, but much of the taproot must be removed or new growth will develop
from the cut surface. Cutting and removing seed heads can stop seed production
in small populations where timely eradication of the plant is not possible
(Pepper 1994).
Mechanical methods: Discing or plowing larger infestations in
wildlands is impractical and not advised. Although it is theoretically possible
to exhaust the carbohydrate reserves of the plantÛªs tuberous roots, this would
require many years of continued effort and several carefully timed passes each
season because artichoke thistle can resprout repeatedly. Discing and plowing
also disturbs the soil, opening it up to reinfestation by this species or other
invaders. The deep root, reaching eight feet (3 m) in depth, also makes
mechanical removal difficult.
Prescribed burning: This could be helpful in removing the
above-ground biomass, making access for chemical control easier and more
efficient. Burning would not be expected to kill the plant, given its perennial
underground storage reserves, but might kill the surface layer of the seedbank.
Applying herbicide to resprouts four to six weeks after they emerge would likely
be most effective for eradicating this thistle from the burned area.
åÊ
|
|
Biological control:
|
Insects and fungi: The plant is a food
source for invertebrates, including earwigs, ants and their aphid consorts,
harlequin beetles, and bees; the latter being found frequently on the flowers.
No USDA approved biocontrol agents exist for this thistle in California. Given
the close relationship of the wild artichoke to the cultivated crop artichoke,
the purposeful introduction of a biocontrol fungus or insect is not likely.
However, Terellia fuscicornis (artichoke fly), an exotic fly from the
Mediterranean Basin, has been discovered, identified, and collected in 1994 from
San Joaquin, Sonoma, and Madera counties. It has subsequently been identified in
wild artichoke populations in a number of other counties. This exotic fly has
been found on both Cynara cardunculus and C. skolymus and feeds on flowers and
seed heads. Since it is a seed-eater, it does not currently threaten commercial
artichoke production. Given the flyÛªs widespread distribution and its potential
as a biocontrol agent on artichoke thistle, a CDFA B rated weed, attempted
extirpation is precluded under agricultural regulations, which is potentially
good news for wildlands (Penrose 1994). By March 1997 the artichoke fly had been
downgraded from a Q to a C listing by the California Department of Agriculture.
The Q listing is a quarantine listing; it cannot be moved around. The C listing,
is a conditional listing, allowing it to be moved if certain conditions are met
with permits between counties (Darling 1997). Until the arrival of the artichoke
fly, herbivory by insects or mammals, including cattle, offered little hope of
controlling artichoke thistle.
Grazing: The spiny nature of the plant deters cattle and sheep
from grazing heavy infestations, but hungry animals will eat the leaves (Parsons
1992). Several species of birds feed on the seeds, but not to any great extent.
Herbivory by deer is not apparent, probably because of the spines. Rabbits
occasionally eat it at the cotyledon stage, but not beyond. The occasional dead
plant encountered in the wild appears to be the work of gophers, but their
impact on the population is insignificant.
åÊ
|
|
Chemical control:
|
Glyphosate is effective in killing artichoke thistle. Cut stump
applications were reported as effective at any stage of growth in nature
preserves in the Santa Monica Mountains (Pepper and Kelly 1994). Subsequent cut
stump application on thousands of plants in Pe̱asquitos Canyon Preserve in San
Diego have confirmed this method (Kelly pers. observation). The cut-stump method
involves cutting the plants as close to the base as possible with a machete,
loppers, or a brush cutter and applying a solution of 25 percent glyphosate (as
Roundupå¨) to the stump. This method is useful for isolated plants or remote
populations where spray equipment is impractical or when it is in close
proximity to sensitive species and foliar spray is not advised. The cut-stump
method has been successfully used to eliminate artichoke thistle from patches of
the endangered San Diego thornmint with no harm to the latter.
A foliar spray of 2 percent glyphosate (as Roundupå¨) can achieve
a kill of 95 to 98 percent on mature, bolting plants. Spraying plants that had
gone to seed also achieved a similar high kill rate. Spraying plants in earlier
stages of growth before the plant sends up its flower stalk kills the
above-ground vegetative structures, but often does not kill all of the roots. In
such cases the plant dies back, but up to 75 percent of sprayed mature plants
resprout in the same season. Transport of fluids is generally up to the stems
and leaves and less down to the roots in pre-bolting plants, probably preventing
sufficient herbicide from reaching the roots. However, spraying seedlings with 2
percent glyphosate is effective. Cutting down dense patches with power tools or
a tractor is a useful prelude to chemical treatment. It allows workers to
penetrate patches with less damage from spines on standing dead plants and
reduces the amount of herbicide needed.
A new herbicide, clopyrlid (as Translineå¨) appears to be
effective when sprayed on this thistle at the rosette stage, but less effective
on mature, bolting plants (Carrithers 1997).
åÊ
|