|
Pennisetum setaceum
|
|
|
|
Scientific name
|
Pennisetum setaceum
|
|
Additional name information:
|
Forsskal
|
|
Common name
|
fountain grass, crimson fountaingrass
|
|
Synonymous scientific names
|
Pennisetum ruppelii, Phalaris setaceum
|
|
Closely related California natives
|
0
|
|
Closely related California non-natives:
|
2
|
|
Listed
|
CalEPPC List A-1,CDFA nl
|
|
By:
|
Jeffrey Lovich
|
|
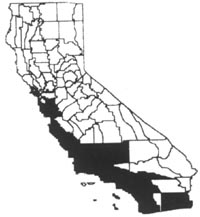
Distribution
|
|
HOW DO I RECOGNIZE IT?
Distinctive features:
|
Fountain grass (Pennisetum
setaceum) is a coarse perennial grass with a densely clumped growth form
and erect stems usually one and a half to five feet tall. The flowerheads are
prominent, nodding, and feathery. They resemble bottlebrushes six to fifteen
inches long, with many, small, light pink to purple flowers.
åÊ
|
|
Description:
|
| Poaceae. Perennial, caespitose grass 16-59 in (40-150 cm) high. Leaves: blades elongate, 0.8-2.6 in (20-65 cm) long, 0.08-0.14 in (2-3.5 mm) wide. Leaf sheath smooth. Several named cultivars, including Û÷Rubrum,Ûª with rose-colored foliage, Û÷Cupreum,Ûª with reddish foliage, and Û÷Atrosanguineum,Ûª with purple foliage (Hammer 1996). Green cultivars reported to be triploid, purple cultivars hexaploid. Purple cultivars may have resulted from chromosome doubling in green cultivar (Simpson and Bashaw 1969). Inflorescence: dense, panicle-like, 3.1-11.8 in (8-30 cm) long. Color varies from light pink to purple. Spikelets on main inflorescence usually 0.18-0.26 in (4.5-6.5 mm) long, about 0.04 in (1 mm) wide; lower glume absent or minute, upper glume generally less than 0.6 times length of spikelet; 2 florets, lower generally sterile, upper fertile; lemmas 3-veined with acuminate tip; paleas absent. |
|
|
|
WHERE WOULD I FIND IT?
|
In California, where fountain grass is
still spreading (RejmÌÁnek and Randall 1994), it is found along the coast from
the San Francisco Bay Area to the South Coast and Baja California. It also
occurs inland in the Sacramento-San Joaquin Delta, southern San Joaquin Valley,
and interior areas of the South Coast and Imperial County. It occurs in
grasslands, deserts, canyons, and disturbed areas along roadsides, especially
adjacent to urban centers. In Hawaii fountain grass invades many types of
natural areas, from bare lava flows to rangelands (Tunison 1992). It has a wide
elevational range, but is limited to areas with a median annual rainfall of less
than fifty inches (
åÊ
|
|
WHERE DID IT COME FROM AND HOW IS IT SPREAD?
|
Originally native to Africa and the Middle
East (Williams et al. 1995), fountain grass has been introduced to many areas,
including Arizona, California, Florida, Hawaii, Fiji, South Africa, and
Australia (Williams et al. 1995). It has spread in large part because of its
popularity as an ornamental plant (Neal and Senesac 1991, Hammer 1996). It is
easily dispersed by vehicles, humans, livestock, and, over short distances, by
wind (Cuddihy et al. 1988), by water, and possibly by birds (Tunison
1992).
The remarkable spread of this species into a broad range of
habitats in Hawaii is attributed to its ability to adapt, physiologically and
morphologically, to different environments. Growth and reproduction traits
change most prominently with altitude, whereas physiological traits are more
strongly affected by physical environment. For example, photosynthetic rates are
higher at higher altitudes (Williams et al. 1995).
åÊ
|
|
WHAT PROBLEMS DOES IT CAUSE?
|
Fountain grass is well adapted to fire
(DÛªAntonio and Vitousek 1992), and plants can recover to pre-burn density, even
increase in density, following a burn. Fire can actually contribute to the
spread of fountain grass (Smith and Tunison 1992). Fountain grass raises fuel
loads, which increases the intensity and spread of fire and results in severe
damage to native, dry forest species adapted to less extreme fire regimes
(Benton 1998). By enhancing the fuel load, fountain grass endangers native plant
communities that are not as fire-tolerant (Tunison 1992). In Hawaii, where it
alters the natural fire regime, fountain grass is a major threat to some
critically imperiled plant species and natural communities (Benton 1998). Fires
fueled by fountain grass impact ground-nesting birds and terrestrial animals as
well.
Thick infestations of fountain grass interfere with regeneration
of native plant species. For example, fountain grass grows faster than the
native Hawaiian grass Heteropogon contortus and can outcompete it in the arid
lowlands of Hawaii. Fountain grass produced 51 percent more total biomass,
allocated 49 percent more biomass to leaves, and had higher net photosynthetic
rates on a leaf area basis than the native Hawaiian grass. Both are C4 species,
and both produced less biomass and allocated more mass to roots in response to
drought. Fountain grass showed no greater flexibility in response to drought
than the native grass. Higher net photosynthetic rates and greater biomass
allocation to leaves, however, gives fountain grass a greater growth rate
relative to H. contortus, which gives it a competitive advantage as an invader
in the arid lowlands of Hawaii (Bruegmann 1996).
åÊ
|
|
HOW DOES IT GROW AND REPRODUCE?
|
Fountain grass can reproduce by either fertilized or unfertilized seeds (Simpson and Bashaw 1969, Dujardin and Hanna 1989). Plants flower from July through October. Fountain grass is apomictic, meaning that it can reproduce asexually by producing seeds from the cells of female plants other than egg cells (Simpson and Bashaw 1969, Dujardin and Hanna 1989). It may also reproduce by seeds produced following pollination and subsequent fertilization of a female egg cell. A green cultivar set approximately 10 percent seed following self-pollination, while a purple cultivar set 0.05 percent. In one study the purple
cultivar set as high as 18 percent seed following application of pollen from Pennisetum ciliare (Simpson and Bashaw 1969). Seeds stored in a laboratory, under dry conditions, for eighteen months decreased in viability from 80 percent to 44 percent (Tunison 1992). Seeds may remain viable in the soil for at least seven years (Tunison et al. 1995). No information is available on conditions favoring germination.
Growth rates of fifty plants with an average basal diameter of
1.1 inch (2.7 cm) were monitored for forty-nine months in Hawaii. Average leaf
length increased 4.6 inches (11.6 cm) a year, and basal diameter increased three
inches (7.74 cm) a year. In five years individual plants can grow leaves up to
four feet long and increase their basal diameter to twelve inches (Tunison,
1992).
|
Fountain grass growing on dunes in Hawaii had vesicular-arbuscular mycorrhizae fungi associated with their roots (Koske 1988).
|
(click on photos to view larger image)
|
|
|
HOW CAN I GET RID OF IT?
|
Fountain grass is difficult to eliminate.
The long-lived seeds make control extremely difficult, and continued monitoring
is essential. Control efforts are most effective when concentrated first on
peripheral or satellite populations to control spread, and then on the core of
the infestation (Tunison et al. 1994). Use of preemergent herbicides may be
useful following removal of an infestation. Surveys on horseback are more
effective than surveys on foot. Helicopter surveys are not efficient, as only
large plants can be spotted, and large infestations of small plants could be
overlooked (Tunison 1992).
åÊ
|
|
Physical control:
|
Mechanical methods: Small infestations of
fountain grass can be removed by uprooting or cutting with weed eaters
(Bruegmann 1996). Heavy tools such as a pick or mattock may be needed to uproot
large plants with a basal diameter over six inches (>15 cm). If
inflorescences are present, they should be cut and placed in plastic bags, then
destroyed to prevent spread of seeds (Tunison et al. 1994). Removal by hand may
need to be repeated several times a year.
Prescribed burning: Fountain grass seems to be stimulated by
fire, so burning is not recommended (DÛªAntonio and Vitousek 1992).
åÊ
|
|
Biological control:
|
No pathogens of fountain grass have been reported in recent
literature (Tunison 1992). Biocontrol efforts would likely also affect related
forage and crop species. The congeneric kikuyu grass (Pennisetum clandestinum)
is a valuable forage grass in some areas (Tunison 1992), and pearl millet (P.
glaucum) is a cultivated species that can hybridize with fountain grass
(Dujardin and Hanna 1989). Fortunately, male hybrids are sterile and females
have poor fertility because of obligate apomixis.
Grazing: In Hawaii cattle eat fountain grass only when no
other grasses are available (Bruegmann 1996).
åÊ
|
|
Chemical control:
|
Extensive infestations of fountain grass
are probably best controlled with the help of herbicides, especially those with
some systemic activity. Ten percent liquid hexazinone (as Velpar) at
Other herbicides are less effective in controlling fountain
grass. Foliar applications of glyphosate (as Roundupå¨) are not consistently
effective (Tunison et al. 1994). Under greenhouse conditions (Neal and Senesac
1991), preemergent herbicide toxicity was greatest using metolachlor granular
application (6.7 kg ai/ha) or spray (6.7 kg ai/ha), napropamide 50 percent
wetable powder (4.5 kg ai/ha), and oryzalin (0.48 kg/l aqueous suspension at 4.5
kg ai/ha). All of these combinations injured and reduced growth of fountain
grass, but did not kill it at the rates used. Not all of these are registered
for use in wildlands in California. Mechanical methods combined with chemical
techniques may be more effective than either method alone.
åÊ
|