|
Conium maculatum
|
|
|
|
Scientific name
|
Conium maculatum
|
|
Common name
|
poison hemlock, carrot fern, poison parsley, spotted hemlock
|
|
Synonymous scientific names
|
none known
|
|
Closely related California natives
|
0
|
|
Closely related California non-natives:
|
0
|
|
Listed
|
CalEPPC List B,CDFA nl
|
|
By:
|
Jennifer Drewitz
|
|
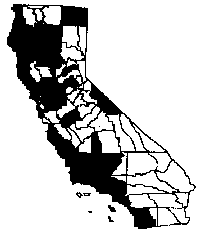
Distribution
|
|
HOW DO I RECOGNIZE IT?
Distinctive features:
|
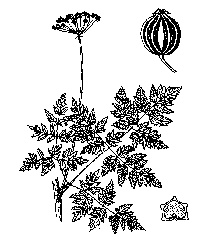
Poison hemlock (Conium maculatum) is a
member of the carrot family. It is usually a biennial, with first-year plants
producing ground-level rosettes. During the second year plants grow from two to
ten feet tall with a stem that is ribbed, hollow, and has purplish streaks or
splotches (Pitcher 1986). Small, white flowers grow in many umbrella-shaped
clusters, each supported by a stalk. Leaves have a somewhat fern-like appearance
and are finely pinnately divided. When crushed, they have a rank odor (Pitcher
1986).
åÊ
|
|
Description:
|
| Apiaceae. Biennial. Stems: hollow, ribbed with purple spots. Leaves: opposite, ovate, tapered, serrate, and finely pinnately divided. Inflorescence: umbels terminal and lateral. There are 4-6 bracts, brown in color, and 5-6 bractlets present. Pedicels abundant. Flower: petals white or yellowish, usually wide with narrow tips. Ray flowers number 10-20, 0.6-2 in (1.5-5cm). Fruit: 0.08-0.12 in (2-3mm) wide, ovate with distinctively wavy ribs (Hickman 1993). |
|
|
|
WHERE WOULD I FIND IT?
|
Poison hemlock has spread throughout
California in areas below 5,000 feet (1,500 m) elevation, excluding the Great
Basin and Desert provinces (Pitcher 1986, Hickman 1993). It is commonly found in
dense patches along roadsides and fields. It also thrives in meadows and
pastures and is occasionally found in riparian forests and flood plains (Goeden
and Ricker 1982). It does best in disturbed areas where soil is moist with some
shade. Poison hemlock is also able to form stands in dry, open areas (Parsons
1992).
|
|
WHERE DID IT COME FROM AND HOW IS IT SPREAD?
|
Poison hemlock is native to Europe, North
Africa, and Asia (Pitcher 1986). It was brought to the United States as a garden
plant sometime in the 1800s and sold as a ÛÏwinter fernÛ (Goeden and Ricker 1982,
Parish 1920). The earliest poison hemlock collections were made in 1893 and 1897
in Berkeley and Truckee, respectively (Parish 1920). Poison hemlock has spread
throughout the United States, Canada, Australia, New Zealand, and South America
(Parsons 1992, Holm et al. 1979).
Poison hemlock reproduces only by seed, which is dispersed by
water, mud, wind, animal fur, human clothing, boots, and machinery (Pitcher
1986, GGNRA 1989). It has no means of vegetative reproduction.
åÊ
|
|
WHAT PROBLEMS DOES IT CAUSE?
|
Poison hemlock can spread quickly after the rainy season in
areas that have been cleared or disturbed. Once established, it is highly
competitive and prevents establishment of native plants by overshading (Serpa
1989). In agricultural areas it interferes with crops and production of feed for
livestock (Jeffery and Robinson 1990).
Poison hemlock is best known for its
toxicity to vertebrates, causing death primarily by respiratory paralysis after
ingestion. The alkaloids in poison hemlock depress the central nervous system.
Symptoms include nausea, vomiting, convulsions, loss of muscle power, dilation
of pupils, slowing of heartbeat, and eventual death from respiratory failure
(Parsons 1992). In livestock, symptoms appear immediately after ingestion, and
death occurs within two to three hours. The recommended treatment is the same as
that for nicotine poisoning: tannic acid followed by a purgative (Parsons
1992).
Poison hemlock is toxic to livestock, wildlife, and humans.
Cattle, sheep, horses, pigs, goats, and fowl are all susceptible to its
toxicity. Poisoning occurs in horses when they eat a quantity approximately
equal to 0.25 percent body weight and in cows, 0.5 percent body weight
(Kingsbury, 1964). Poisonings are usually not caused by direct foraging of
poison hemlock, but by consumption when it is mixed in stock feed (Parsons and
Cuthbertson 1992).
Wildlife is also susceptible to the toxic effects
of poison hemlock. Ten percent of an elk population on
Grizzly Island, California, died from ingesting poison hemlock in 1985 (Parsons
and Cuthbertson 1992).
Field experiments have not established any allelopathic effects
of poison hemlock (Serpa 1989).
Ancient Greeks used poison hemlock to carry out judicial
executions, including the execution of Socrates (Parsons and Cuthbertson 1992).
Most human poisonings occur when the leaves are eaten by people confusing them
with edible seeds used as spices, or when children use the hollow stems as
flutes (Kingsbury 1964). Seeds and young leaves are the most toxic parts of the
plant. It is recommended that gloves be worn when handling the plant, as some
people develop dermatitis, and that inhalation of particles be minimized
(Parsons and Cuthbertson 1992). Children should be monitored when in areas
containing poison hemlock.
åÊ
|
|
HOW DOES IT GROW AND REPRODUCE?
|
Reproductive plant parts develop in mid-April, usually one year after germination (Amme 1988). In summer, once plants have set seed, they dry up and die leaving tall stalks to shade out other plants. The seed of poison hemlock is fully developed by mid-June. Plants disperse about 90 percent of their seed in September through December, with the remainder dispersed by late February (Baskin and Baskin 1990). This lengthy dispersal period allows poison hemlock to produce new seedlings continuously for several months (Baskin and Baskin 1990).
|
Poison hemlock has a large range of conditions in which it can germinate. It can germinate at temperatures greater than 9.4 C and lower than 33.8 C. It can germinate in darkness as well as in light. About 85 percent of seed produced is able to germinate as soon as it leaves the parent plant (Baskin and Baskin 1990). The remainder is dormant and requires certain environmental conditions (thought to be summer drying) in order to germinate (Baskin and Baskin 1990).
|
(click on photos to view larger image)
|
This ensures that some seed will remain in the seedbank until
the following growing season. Seed can remain viable in the soil for up to three
years (Baskin and Baskin 1990). It germinates most readily in soil, but can also
germinate in sand. The combination of long seed dispersal period, seed dormancy,
and non-specific germination requirements enable poison hemlock seedlings to
emerge in almost every month of the year (Roberts 1979). Germination takes place
in all months of the year except April, May, and July, with late winter and
early spring being the periods of greatest germination (Roberts 1979). Most
vegetative growth occurs in winter months, with plants developing a deep taproot
that is sometimes branched (Pitcher 1986).
åÊ
|
|
Physical control:
|
Manual methods: Hand pulling of poison hemlock is effective,
especially prior to seed set, and easiest when the soil is wet (Parsons 1992).
Because of the biennial nature of the plant, the entire root system does not
need to be removed (Pitcher 1986).
Mechanical methods: Spring mowing has proven effective in
killing mature plants, yet regrowth may occur and new seedlings may continue to
establish (Amme 1988). A second mow in late summer is recommended to eliminate
remaining or subsequent growth (GGNRA 1989). Because poison hemlock seed has
been shown to germinate up to three years after dispersal, a third year of
mowing may be necessary (Baskin and Baskin 1993).
Prescribed burning: Joseph DiTomaso, with the University of
California, Davis, Weed Science Group, states that burning is probably not a
good control option. In areas where poison hemlock is the dominant vegetation
(usually moist environments), sufficient dried material would not be available
to provide adequate fuel to control poison hemlock before fruit maturation. This
method has yet to be tried on poison hemlock.
åÊ
|
|
Biological control:
|
Insects and fungi:
Although biocontrol is being examined, at this time there is no USDA approved
biocontrol agent for use on poison hemlock in California. Goeden and Ricker
(1982) found few insects attacking poison hemlock in California. Those that were
found feeding on poison hemlock were unspecialized and polyphagous species
(Goeden and Ricker 1982). In 1994 Berenbaum and Harrison published a paper
noting a native European oecophorid caterpillar, Agonopterix
alstroemeriana, was infesting poison hemlock in northeastern and western
parts of the United States (Berenbaum and Harrison 1994). According to Berenbaum
and Passoa (1983), ÛÏC. maculatum appears to be the sole host of A.
alstroemeriana.Û It is not known how A. alstroemeriana
was
introduced into the United States, but this defoliating moth is now described as
a biological control agent providing good to excellent control when present in a
medium to heavy infestation in the Pacific Northwest (William et al.
1996).
|
|
Chemical control:
|
Effective post-emergent herbicides include
2,4 D ester, 2,4 D amine, and glyphosate plus surfactant, all to be applied in
late spring. 2,4 D has been effective when sprayed at 1.0 lb ai/acre (1.1 kg/ha)
and mixed with a wetting agent (Jeffrey and Robinson 1990). Glyphosate at a rate
of 1.0 lb/acre (1.1 kg/ha) plus surfactant (as Roundupå¨) has also proved
effective in killing poison hemlock, especially in the rosette stage (Amme 1988,
Jeffery and Robinson 1990). Glyphosate plus surfactant (as Roundupå¨) at a rate
of 1.0 lb/acre (1.1 kg/ha) has also proved effective in killing poison hemlock,
especially in the rosette stage (Amme 1988, Jeffery and Robinson
1990).
åÊ
|