|
Lythrum salicaria
|
|
|
|
Scientific name
|
Lythrum salicaria
|
|
Additional name information:
|
L.
|
|
Common name
|
purple loosestrife
|
|
Synonymous scientific names
|
none known
|
|
Closely related California natives
|
15
|
|
Closely related California non-natives:
|
0
|
|
Listed
|
CalEPPC List B
|
|
By:
|
Carri Benefield
|
|
Distribution
|
Listed as noxious by 16
states
|
|
HOW DO I RECOGNIZE IT?
Distinctive features:
|
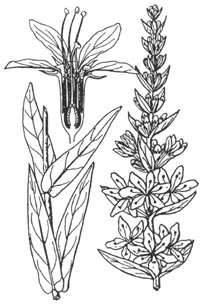
Purple loosestrife (Lythrum salicaria) is a
perennial wetland herb, typically less than five feet tall with showy spikes of
reddish purple flowers. Each flower has five to seven pink-purple petals
surrounding a small yellow center. Plants enlarge at their bases each year with
more stems, sometimes becoming a rounded bush-like clump of thirty to fifty
stems arising from a single root stock. Leaves are long and narrow, opposite to
whorled, and closely attached to the four-sided stem.
Purple loosestrife may be distinguished from similar looking
species by its generally larger size, opposite to whorled leaves, and less
widely spaced flowers. It may be confused with other native species such as
fireweeds, vervain, or the closely related Lythrum californicum, so accurate
identification should be made before control measures are attempted. Fireweed
(Epilobium angustifolium) has spikes of magenta flowers, but these spikes are
narrower (four to five inches wide at the base) and more conical. Fireweed stems
are round, and the leaves are alternate. Blue vervain (Verbena hastata) has
smaller purple flower spikes, and the edges of the leaves are toothed. L.
californicum has smaller, narrower leaves and half (six versus twelve) the
number of stamens.
åÊ
|
|
Description:
|
| Lythraceae. Perennial herb. Stems: 1-5 ft (0.3-1.5 m), occasionally to 10 ft (3 m) tall, erect stems; four-sided, smooth to lightly hairy, woody with age, persisting through winter. Leaves: opposite or whorled, 2-6 in (5-14 cm) long, generally two-ranked, lanceolate to narrowly oblong, edges smooth, sessile (Hickman 1993). Inflorescence: terminal, spike-like, composed of numerous flowers closely arranged in leaf axils of ascending flower-bearing branches (Rawinski et al. 1982). Flower: trimorphic in regard to the relative lengths of stamens and style; hypanthium cylindric; sepals |
|
|
|
WHERE WOULD I FIND IT?
|
Purple loosestrife can be found in
scattered freshwater wetlands in northern and central California. Counties with
infestations include Humboldt, Mendocino, Modoc, Shasta, Siskiyou, and counties
in the Sacramento Valley and San Francisco Bay Area. Other infestations occur
along rivers in Fresno and Kern counties, such as along the Kern River in the
southern Sierra. It is also widespread in the eastern United States. It is
common in disturbed wetland habitats, such as stream and river banks, edges of
ponds, lakes, and reservoirs, flooded areas, ditches and roadsides, but it can
colonize fairly pristine wetland areas, including marshes, wet prairies,
meadows, pastures, and bogs. While loosestrife generally grows best in full sun
in moist, organic soils, it can tolerate a wide range of soil moisture regimes
(intermittent or continuous flooding up to 1.25 feet or 0.4 m), soil types
(clay, sand, silt, and muck; pH 4-9), nutrient levels, temperature, and light
(up to 50 percent shade) (Skinner et al. 1994). If water levels drop,
loosestrife can easily become established, and populations can expand on exposed
soils.
åÊ
|
|
WHERE DID IT COME FROM AND HOW IS IT SPREAD?
|
Purple loosestrife is native to Eurasia and
was introduced into the northeastern United States in the early 1800s in shipsÛª
ballast, as an herbal, as an ornamental plant, and by beekeepers. It spreads
primarily by seed, but can grow from broken-off stem and root segments and can
resprout from the root crown (Bender et al. 1987). Seed dispersal occurs
primarily by wind and water (most rapidly by flowing water, but also by
permanent standing water), and in mud attached to wildlife, boats, vehicle
tires, and footwear (Heidorn 1990, Skinner et al. 1994).
åÊ
|
|
WHAT PROBLEMS DOES IT CAUSE?
|
Purple loosestrife is a hardy perennial
that can rapidly degrade wetlands, diminishing their value for wildlife habitat.
Once established, it forms extensive monotypic stands that displace native
vegetation relied on by wetland species for food and habitat (Nelson and
Getsinger 1994, Bender 1987). Infestations jeopardize the federally listed
endangered bog turtle (Clemmys muhlenbergi) among other species, though not in
California. Purple loosestrife also clogs waterways and wetlands used for
boating and other recreational activities. Impacts on agriculture include
changes in hydrology and soil conditions of wetland pastures and meadows and
clogged irrigation systems. Purple loosestrife has the potential to infest rice
fields. An estimated 190,000 hectares of wetlands, marshes, pastures, and
riparian meadows are affected in North America each year, with an economic
impact of millions of dollars.
åÊ
|
|
HOW DOES IT GROW AND REPRODUCE?
|
Purple loosestrife flowers from late June through September. Seed set begins in mid-July and continues into late summer. New shoots arise the following spring from buds at the top of the rootstocks. Long-lived seeds are copiously produced (more than two million seeds per mature plant) and shed gradually from capsules through the winter, contributing to an immense seedbank. It has not been determined how long purple loosestrife seeds remain viable, but it is thought to be at least several years.
|
Most seedlings establish in late spring to early summer and produce a floral shoot up to one foot (30 cm) high the first year. Moisture is the most critical habitat requirement for seed germination. Critical temperatures for germination range from 60 to 72 degrees F (15-20 degrees C) (Skinner et al. 1994).
|
(click on photos to view larger image)
|
|
|
HOW CAN I GET RID OF IT?
|
The best method for removal of purple
loosestrife depends on the site, the age and size of the infestation, the
importance of impacts to non-targeted species, and the type and quantity of
resources available to gain control. Ideally, an integrated pest management
(IPM) approach should be developed to take advantage of the best available
control methods. All methods require appropriate timing and follow-up monitoring
to be successful.
In natural communities with no known invasion, prevention and
early detection are the best approaches. Potential habitat should be searched
annually for newly established populations in late July through August, when
blooming plants are easily spotted from a distance. In addition, public
education efforts and avoidance of unnecessary disturbances of wetland habitat
are desirable (Heidorn 1990, Mountain 1994).
åÊ
|
|
Physical control:
|
Manual methods: In areas with individual
plants and small localized stands (up to 100 plants), younger plants (one to two
years old) can be pulled by hand. Plants should not be pulled after flowering,
because this will scatter seed. Older plants, particularly those in loose soils,
can be dug out by teasing roots loose with a cultivator, but great care should
be taken to bag and remove all plant parts from the site because broken-off
pieces can re-root (Heidorn 1990, Skinner et al. 1994). Dispose of plants by
burning (preferable) or by drying.
Several methods have been shown to be ineffective and should be
avoided: Cutting at water level and below water level has resulted in
resprouting and reestablishment. Mowing, if timed correctly, has been shown to
prevent flowering and seed set and reduce carbohydrate reserves, but it can
result in dissemination of seeds and re-rooting of stem fragments (Heidorn
1990). Flooding is ineffective unless plants can be inundated throughout the
summer, but this is detrimental to most other emergent species and promotes
spread of loosestrife to shallow areas (Skinner et al. 1994). Disking and
flooding has shown some success, but most areas cannot be drained and dried
enough to support heavy equipment (Skinner et al. 1994). Burning does not kill
buried rootstocks, which resprout later. Herbicide plus burning was attempted
without success (Skinner et al. 1994).
åÊ
|
|
Biological control:
|
Insects and fungi: Since the 1980s the U.S.
Fish and Wildlife Service and the International Institute of Biocontrol have
been screening, testing, and importing natural purple loosestrife enemies. In
the early 1990s the USDA and Washington and Oregon state departments of
agriculture gained approval for the release of five agents, three weevils and
two leaf eating beetles (Galerucella spp.). California required extra
testing (for potential impacts on crape myrtle, a widely used ornamental) before
obtaining a permit in 1998 to conduct test releases of leaf-eating beetles in
California. Considerable damage to purple loosestrife from the leaf-eating
beetles has been reported in other states and in California test release sites.
However, further research is necessary before approval of large-scale release of
Galerucella spp. in California. Other states have reported that it can
take four to six years for Galerucella spp. to become fully
established.
Plant competition: Research on reseeding with competitive
species is still in its early stages. It has been difficult to find species that
compete successfully with purple loosestrife in a wide variety of conditions and
that are not themselves invasive. Japanese millet plantings in Minnesota have
been successful in the short term, but this non-native species regenerates
poorly and must be replanted every year (Bender et al. 1987).
åÊ
|
|
Chemical control:
|
In areas up to four acres with clusters in excess of 100 plants,
the most effective herbicides available are glyphosate (as Roundupå¨ and Rodeoå¨)
applied in spot applications. Glyphosate is non-selective, so care should be
taken to avoid contact with non-targeted species that are critical in
recolonizing the site and inhibiting loosestrife reestablishment (Bender 1987,
Skinner et al. 1994). Herbicides should be applied to the foliage during the
blooming season (late June to early September). Effective applications have been
reported at both flower initiation (usually late June) (Heidorn 1990, Nelson and
Getsinger 1994) and after peak bloom (usually late August) (Balogh 1986,
Rawinski 1982). Timing of application is essential for maximizing chemical
effectiveness and preventing seed production. Glyphosate should be applied by
hand sprayer as a 1.5 percent solution (2 oz/gal water). Rodeoå¨, registered for
aquatic use, should also be applied as a 1.5 percent solution, with the addition
of a surfactant approved for use over water, as specified on the label (Heidorn
1990).
2,4-D approved for aquatic use is the second most commonly used
herbicide, despite its inconsistent ability to control purple loosestrife. This
herbicideÛªs selectivity for broadleaf plants and low cost make it appealing.
Applied before flowering (late May to early June), it is most effective in
controlling first-year seedlings and preventing seed production in mature
plants, but it does not kill the plants and many will likely resprout (Bender
1987; Skinner et al. 1994).
Triclopyr (as Garlon 3Aå¨or Renovateå¨) is not not yet approved
for aquatic use, but it has great potential because it is selective for
broadleaf plants and does not harm grasses and most other monocots, which are
important in wetland habitats. While some reports point to the inconsistency of
Garlon 3Aå¨ in the field (Katovich et al. 1996), others indicate that it is
effective and promotes regrowth by grasses and other wetland monocots (Nelson
and Getsinger 1994). Garlon 3Aå¨ continues to undergo evaluation for its use in
aquatic environments.
åÊ
|